Solvency offers great solutions
High solvent power is the secret of the success of naphthenic oils in chemical applications. Anna Eriksson, Marketing Manager, Nynas Naphthenics, offers a couple of examples that explain why.
 Anna Eriksson
Anna Eriksson
Marketing Manager, Naphthenics
Nynas AB
The excellent solvency performance of naphthenic process oils gives Nynas a competitive edge in many of the around 20 different application areas the company serves within the chemical industry segment.
“Solvent power is important in many different applications. One example is the dissolving of resins in printing ink formulations. We have tested Nynas naphthenic oils of varying viscosity alongside Group I paraffinic oil of matching viscosity and found clear correlations to their respective solvent power,” says Anna Eriksson.
The screening was conducted using cloud point evaluation. The cloud point is the temperature at which dissolved solids are no longer completely soluble in a fluid. The solvent power is responsible for keeping the resin in solution.
“The cloud point can also be used to indicate the time and energy needed for the dissolution, and this can be used to assess the economic and environmental impact of using naphthenic versus paraffinic oil. The high solvent power of naphthenic oil makes it a clear winner.”
Another example of the importance of solvency can be found in antifoam production.
“When an antifoam is prepared, the active component must be dissolved and kept in solution until it is needed. Comparative studies clearly show that oils with higher solvent power facilitate the dissolution of active ingredients such as those used in antifoams,” says Anna Eriksson.
The solvent power plays an important role in all production processes where oil is used. High solvent power offers formulators flexibility to use a wide range of raw materials, higher loading of active ingredients or additives, and shorter process times.
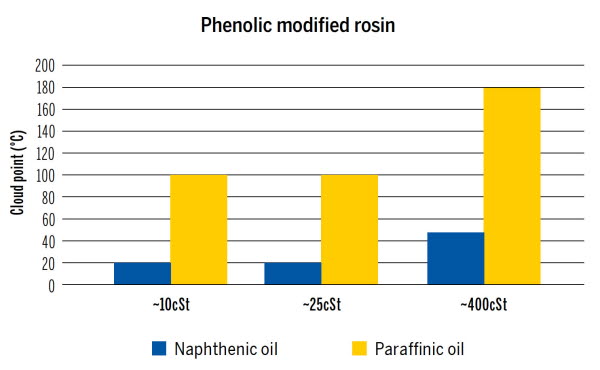
In a comparative study, 10 % of phenolic rosin ester resin in oil was heated until fully dissolved, then allowed to cool until precipitation (a second phase) occurred. The high solvent power of the naphthenic oil has a great impact.
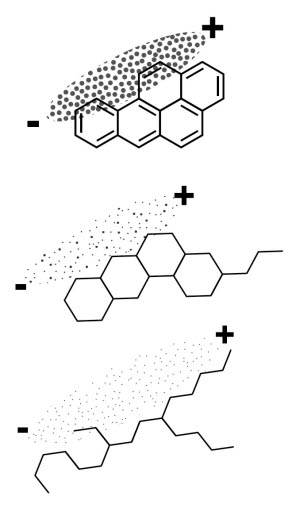
- The solvent power of an oil is dependent on its dispersive forces. These, in turn, depend on the electron density which depends on the oil’s molecular structure and bond type.
- Aromatic (unsaturated cyclic) hydrocarbons (a) display very powerful dispersive forces, due to very high electron density. However, the unsaturation can also promote unwanted reactions.
- Stable and non-harmful, naphthenic (saturated cyclic) structures (b) have stronger dispersive forces than paraffinic (saturated straight) structures (c).
- The stronger the dispersive forces, the higher the solvent power.













































